| Monomers |
|
| Monomers are small molecules that can link end to end to form long polymers.
This reaction is known as polymerisation.
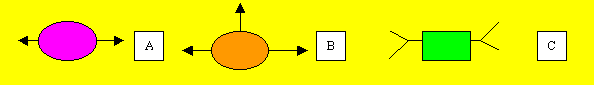
Monomers have two or more sites where joining can occur. Consider the monomers below. |
|
|
Consider the polymerisation of "A" and "C" |
|
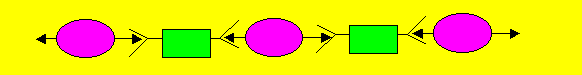
| In this instance a linear(straight) polymer is formed. Linear polymers
pack together to form a thermoplastic. Thermoplastics are plastics that
melt when heated. Consider the polymerisation of "B" and "C" |
|
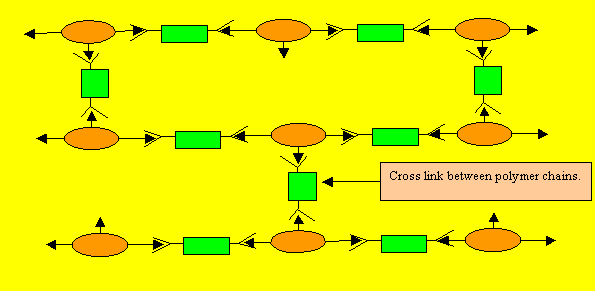
| Notice how the polymers are
linked to one another. Thermosetting plastics
can not be softened with heat. The choice of a monomer is crucial in producing the right polymer. Any monomer with more than two joining sites can produce thermosetting plastics. Thermosetting plastics are also known as cross-linked polymers. |
|
The creation of a thermosetting plastic is shown on the right. Notice how the monomers are held tightly in position by strong links in three dimensional space. This creates a plastic that is heat resistant and does not melt. Cooking pot and frying pan handles are made of such a plastic.
|
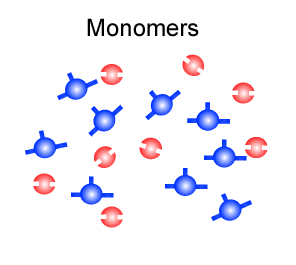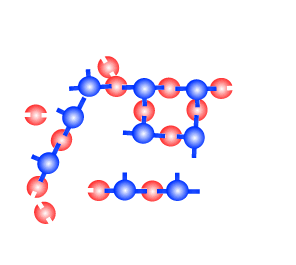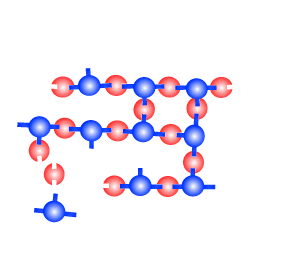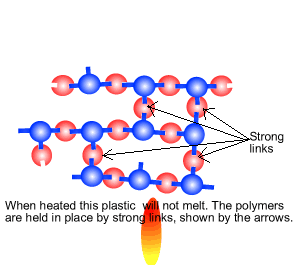 |
| As you can see from the video on the right, thermosetting plastics are heat resistant. |
|
| It is worth noting that thermosetting plastics have polymers that grow in 3D space, as the animation on the right depicts. | 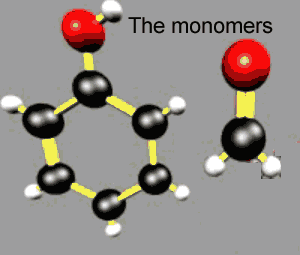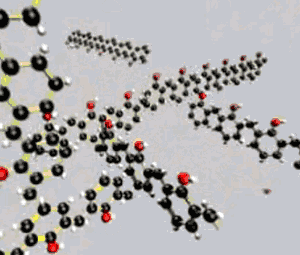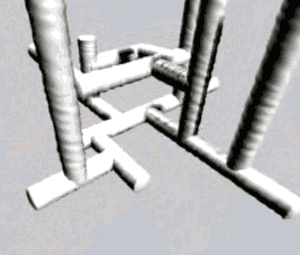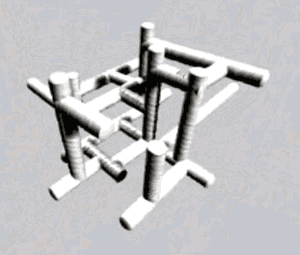 |
| Monomers are chosen carefully so that the final product, plastic, has the desired properties. Below are some examples. | |
| PVC or poly vinyl chloride is a plastic used for a large range of applications some of which include pipes. The monomer is chloroethene. By replacing a hydrogen on the ethene molecule with a chlorine the intermolecular forces, namely, dipole-dipole and dispersion forces are significantly increased between the polymer molecules. |  |
The lightweight, checkout bags, pictured on the right, are made from high density polyethylene (HDPE). The heavier, tougher plastic bags found in boutique and department stores are made from low density polyethylene (LDPE). Both HDPE and LDPE are made from ethene monomers, however, under certain conditions, long straight polymers are formed of polyethene that pack closely together and hence form HDPE. When the conditions are changed the polymers become branched and can not pack closely together and hence form LDPE. |
 |
| Nylon is a polymer made from two different monomers. The major intermolecular force acting between the polymers is hydrogen bonding. This type of intermolecular bonding is relatively strong, hence the strength of the nylon ropes. |  |